
EXERCISE 1
NON-SPECIFIC HOST DEFENSE
I. INTRODUCTION
Non-antibody humoral factors, enzymes, and phagocytosis are part of the
NON-SPECIFIC antimicrobial systems which assist in defense against infection and
disease. Examples include intact skin, ciliary action in the lung, antibacterial
substances in secretions, opsonic factors in serum and phagocytosis.
A
host's initial response to the introduction of foreign material is the acute
inflammatory response. When this involves bacteria, and the numbers are small,
the ensuing inflammatory response may resolve the infection; involvement of the
ACQUIRED resistance mechanisms - humoral and cellular immunity - may not be
necessary.
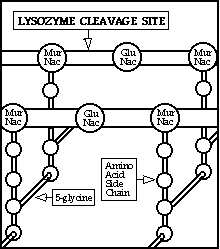
External secretions and neutrophil granules contain many
substances with antibacterial activity, including lysozyme. Lysozyme (or
muramidase) can cleave the mucopeptide layer of most non-encapsulated
Gram-positive bacterial cell walls resulting in cytoplasmic blebbing of the
bacterial cell through the wall defect. If the external osmolality is
sufficiently low, direct lysis can occur, or hydrophobic proteins with
detergent/pore forming capacity derived from neutrophil granules may act on the
exposed bleb and cause lysis.
The complement system comprises about 10%
of normal serum proteins with C3 component present in the greatest amount. A
number of important protective/ inflammatory functions are mediated by activated
complement factors, in particular, lysis. The mechanism of complement cell lysis
is not clearly defined. Activation of complement fragments on the cell surface
may lead to the assembly of the terminal membrane attack complex (C5-9) and the
formation of a transmembrane pore that leads to Na/K pump dysfunction and osmotic
lysis. Both eucaryotic and procaryotic cells are sensitive to this method of
killing. 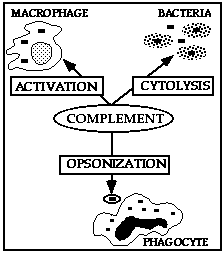
Enhanced phagocytosis is
often the ultimate effector mechanism in both nonspecific and specific immunity,
whether humoral or cellular. Whole blood contains cells (neutrophils, monocytes)
that are potentially phagocytic. The native rate of phagocytosis by these cells
is low. Treatment of a foreign particle with normal serum opsonin (C3b derived
from alternate complement pathway activation by bacterial cell surface
polysaccharides) normally promotes phagocytosis.
| Mechanism | Factor |
How entry of
microorganisms
is limited: | Keratin layer of intact skin
Lysozyme
in tears, etc.
Fatty acids of the skin
Respiratory cilia
Normal flora
of throat, colon and vagina
Low pH of vagina and stomach |
How growth of
microorganisms
in the body is
limited: |
Natural killer cells
Phagocytes
Interferons
Transferrin and
lactoferrin
Complement
Elevated body temperature (fever)
Inflammatory
response |
II. LAB WORK
A. COMPLEMENT
ACTION
Materials supplied: (work in pairs)
- 1 tube of
COMPLEMENT on ice (green cap)
- 1 tube of HEAT-INACTIVATED COMPLEMENT
(red cap)
- 2 tubes of Gram-negative BACTERIA (brown caps)
- 2
cotton swabs
- 4 pipets, 1 ml
- 2 TS Agar plates (TSA)
- 1 PipetAid (lab
drawer)
Procedure: - Use the blue PipetAid (lab drawer)
to pipet 0.2 ml of complement (green cap ) into 1 tube of
bacteria. Label it and one TSA plate, "Active C".
- Use a new pipet
to pipet 0.2 ml of heat-inactivated complement (red cap ) into the
other tube of bacteria. Label it and the other plate, "C Control".
- Incubate the two bacteria tubes in a WATERBATH at 37ˇC for 30 min. While
this incubates, complete Part B. Lysozyme Action.
- After the incubation,
retrieve the bacteria tubes from the waterbath, and agitate each tube, mixing
well. Pipet 0.2 ml from "Active C" tube and deposit it on the
center of the first TSA plate. Discard the pipet directly into the pipet
jar. Use a sterile cotton swab to spread the liquid evenly over the surface
of the TSA plate. Discard the swab directly into the small, red Biohazard
bag. Throw the swab wrapper into the trash.
- Use another sterile pipet
to remove 0.2 ml from the "C Control" tube and deposit the liquid on the
center of the second TSA plate. Discard the pipet directly into the pipet jar.
Use a sterile cotton swab to spread the liquid evenly over the surface of the TSA
plate. Discard the swab directly into the small, red Biohazard bag.
- Invert the plates, label with your name, and place them in the 37C
air incubator. At the next lab period, compare the number of colonies on the two
plates and note the non-specific bactericidal effect of complement.
Photo of Complement Lysis Results
B. LYSOZYME
ACTION
Materials supplied: (work in pairs) - 1 tube of
LYSOZYME on ice (yellow cap )
- 1 tube of SALINE (black cap )
- 2 tubes of BACTERIA (blue caps )
- 2 pipets, 1 ml
- 1 tube of
DETERGENT (purple cap )
- 1 Pasteur pipet
- 1 PipetAid (lab drawer)
- 1 rubber bulb (lab drawer)
Procedure: - Label one
tube of bacteria, "Control". Use a 1 ml pipet and the blue PipetAid, to
transfer 0.1 ml of saline to this tube.
- Label the other bacteria tube
"Lysozyme". Use the other 1 ml pipet to transfer 0.1 ml of
lysozyme to this tube.
- Mix each tube well and allow the lysozyme to react
for 3 minutes. Warm the tubes between your hands.
- Use a Pasteur pipet and
rubber bulb to put 1 drop of detergent into both tubes. Mix.
- Examine for the clearing of turbidity. This clearing indicates lysis due to
enzymatic digestion of the bacterial cell wall. Use the saline tube for
comparison.
Photo of Lysozyme Lysis Results
While every effort is made to ensure that this information is up-to-date and accurate, the statements found on this page are for informational purposes only.LSUMC WWW Publication Policy, 9/23/96




