
EXERCISE 4
ANTIGEN-ANTIBODY II
I. INTRODUCTION
D.
AGGLUTINATION AND IMMUNE LYSIS
Agglutination is a two-step reaction. The
antibody is in solution so the character of the reaction depends largely on the
physical state of the antigen. Antigens in a particulate state (bacteria
or erythrocytes) are clumped or agglutinatedby the soluble antibody (agglutinin).
The PRIMARY reaction is the binding of the Fab portion of antibody to
antigenic determinants on the particulate antigen and so is not visible to the
eye.
The SECONDARY reaction is the result of the cross-linking by
multivalent antibodies, of the particles of multivalent antigen to form
aggregates largeenough to be detected visually.
The foregoing two-component
system can in certain instances be enhanced by the addition of a third component,
complement (C'). This is exemplified by the reaction of immune
hemolysis, in which the antibody " sensitizes" erythrocytes
which are then lysed by complement. Other mammalian cells (e.g., leukocytes) and
certain bacteria (e.g., Gram-negative organisms) can be lysed by this system.
The most obvious clinical application of agglutination is
blood-typing; erythrocyte blood group surface antigens (e.g., the ABO blood group
factors) react with antibody that is specific for these allo-antigens to produce
agglutination. The cross- matching of blood depends upon this technique.
The
surface antigens of bacteria also react with appropriate antibodies to produce
agglutination. A patient's serum can be tested for the presence of
antibody to known antigens of certain bacteria (e.g., Salmonella typhi), giving
diagnostically significant information. Conversely, bacteria such as Shigella,
Escherichia, or Proteus isolated from cultures of clinical specimens are
identified most definitively using antibodies of known specificity (serotyping).
E. BACTERIAL OPSONIZATION
Certain bacteria, such as
Streptococcus pneumoniae and Hemophilus influenzae, can by virtue of their slimy
capsules evade phagocytosis, thus prolonging disease. If antibody specific for
this capsule is present in the patient's serum, it will "
coat" the organism making it more " sticky" and thus more easily phagocytosed. Activated complement components on the surface of
cells also aid in phagocytosis. Substances involved in this type of reaction
are referred to as opsonins . II. LAB WORK
A. BACTERIAL AGGLUTINATION
Materials supplied: (work in pairs)
- 1 tube of unknown ENTERIC BACTERIA with dye added (brown cap )
- 1 tube of SALINE (pink cap)
- 1 Agglutination slide
- 2 Pasteur
pipets
- Salmonella antiserum in dropper bottles
- Shigella antiserum in
dropper bottles
Procedure: - Label the three wells on the
Agglutination slide: Salmonella, Shigella and Control.
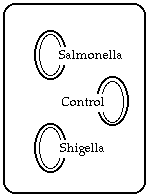
- Place a drop of Salmonella antiserum in one well, and a
drop of Shigella antiserum in the other well. Use a Pasteur pipet to place a
drop of saline in the third well as Antigen Control. Discard the pipet properly.
- Use the other Pasteur pipet to add one drop of the unknown bacterial
suspension to each of the three wells. Discard the pipet properly.
- Rock the
slide gently to achieve mixing of the cells and antisera. Let sit for about 2
minutes, mixing occasionally.
- Read both experimental wells for agglutination:
the suspension appears granular. Use the saline well as a comparison (Negative
Control). Examine your slide with the dissecting scope (front table) to confirm
your conclusions. Discard these slides in the specifically marked disposal
bucket.
- Record your results in the slide diagram on
the Report Sheet, as + or - agglutination. Identify the unknown enteric
bacteria.
- Observe the demonstration on blood typing.
B. ERYTHROCYTE TUBE AGGLUTINATION Materials supplied: (work in
pairs)
- 1 metal rack with 10 tubes.
- 1 tube of complement fixation
(CF) saline for making dilutions. (yellow cap)
- 1 tube of erythrocyte
antigen (0.75% suspension of sheep RBC)
- 2 pipets, 5 ml
- 1 pipet, 1 ml
- PipetAids (blue/green, in lab drawer)
Procedure: Note:
Tube #1 is already numbered and contains exactly 0.5 ml serum (heat
inactivated).
- Be sure all tubes are numbered 1-10. Label tube 1
with your name.
- Use the green PipetAid and one 5 ml pipet to deliver exactly 0.5 ml CF saline
to each of the 10 tubes.Discard this pipet. Put tube 10 aside.
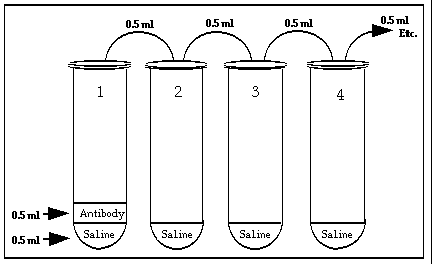
- Use the 1 ml
pipet and the blue PipetAid to mix the contents of tube 1 and transfer 0.5 ml to
tube 2. Mix the contents of tube 2, withdraw 0.5 ml and transfer it to
tube 3. Perform this same action on each of the tubes through tube 9. Mix the
contents of tube 9, withdraw 0.5 ml and DISCARD BOTH in the pipet jar.
Tube 10 will be the ANTIGEN CONTROL.
- Resuspend the sheep RBC suspension and
use the other 5 ml pipet and the green PipetAid to add exactly 0.5 ml of
erythrocyte suspension to each tube 1-10.
- .Shake the rack well to
mix the reagents, and place in the 37C WATERBATH. Take care that water does
not get into your or other tubes. After 1 hour, all racks will be removed by
the instructor and stored in the refrigerator until the next lab period.
Next Lab Period Do not shake or mix your tubes.
Start with Tube 10 and look at the pattern the RBCs make on the tube bottom.
This is called a " button" . Compare all of the other tubes
to this Control tube.Determine the titer by finding the tube with the highest
serum dilution (lowest antibody concentration) that has an agglutination pattern
that is completely different from the Control.The reciprocal of this
dilution is expressed as the TITER. Intermediate reactions appearing between the
Control tube and this other tube can be graded as " +/-" or
" partial" agglutination. (See example at front table)
- Observe the demonstration of immune hemolysis at
the front table. The antiserum was diluted out serially in the same manner as
you performed in lab. Active complement (C' ) was added to tubes 1- 9,
and the C' Control tube. The same RBC suspension used in lab was
added, the solutions mixed and incubated for 30 minutes at 37C, then refrigerated
overnight.
- Notice that the titer obtained by the Complement assay is higher than
that determined by your experiment.
Agglutination and Hemolytic Results
| Tube # | 1 | 2 | 3 | 4 | 5 |
6 | 7 | 8 | 9 | 10 |
| Serum
dilution | 1:2 | 1:4 | 1:8 | 1:16 |
1:32 | 1:64 | 1:128 | 1:256 | 1:512 |
Control |
Agglutination
End Point |
|
|
|
|
|
|
|
|
|
|
Hemolytic
End Point |
|
|
|
|
|
|
|
|
|
|
Why is this, if the same
serum was used in each assay?
Record the hemolytic and agglutination endpoints
here and on your Report Sheet.
Hemagglutination
Results
C. NEUTRALIZATION OF BIOLOGICAL ACTIVITY BY
ANTIBODYBacterial Opsonization:
- Observe the two slides of phagocytosis on demonstration at the front
table. One shows pnemococci without serum, and the other shows pneumococci with
specific anti-capsular antibody.
- The specific anti-capsular antibody binds to
the capsular material on the cell, becoming an Ag-Ab complex, activating
complement that enhances the phagocytic capacity of macrophages and
lymphocytes.Many more pneumococci can be observed within the cells in the sample
with the opsonized bacteria.
Neutralization of Virus Receptors:
- The serodiagnosis of a viral infection is dependent upon the reaction of
virus-specific antibody with either virus or virus antigen. In order to assure
that the positive reaction seen between virus and antibody is relevant to the
current disease (and not a long standing immunity), a serum sample must be
obtained as early in the course of disease as possible (acute phase serum), and a
second sample collected in the later stages (convalescent phase serum). The
acute and convalescent sera can then be compared for an increase in the amount of
specific antibody to the suspected virus. An increase in titer greater than
fourfold is considered indicative of a specific immune response, and thus
provides serodiagnosis. Fourfold increases are significant because dilution
errors would not transcend more than one dilution tube (twofold).
- In the
example demonstrated at the front table, Influenza A virus by itself can
agglutinate RBCs because it recognizes a receptor on the RBC surface. If the
virus is first mixed with antibody to the virus, then the receptors will
be blocked and the virus will be " neutralized" and will be
unable to agglutinate the RBCs. This reaction is called hemaagglutination
inhibition. The quantity of virus in this test is constant so one can quantitate
by dilution, the amount of Influenza A antibody in the serum.
- On display at
the front tables are two racks of tubes, each representing the quantitation of
Influenza A antibody in a serum sample. The serum samples differ only as to when
in the course of the disease they were obtained.
- Determine the titers of each
serum sample in the same manner as you did with your own tubes, by comparing each
tube with the controls. Notice that a lack of hemagglutination (i.e. a
button) indicates a positive antigen-antibody reaction. Make sure you
understand the difference between this test and the hemagglutination test you
performed in lab.
The Virus Control shows you the pattern of
hemagglutination when no antibody is present , and proves that the virus
is capable of agglutinatingRBC' s.
The Antibody Control
shows you the pattern of hemagglutination when no virus is present, e.g.,
when it is all neutralized .
The Cell Control shows you that the
other patterns weren't influenced by aberrant cell settling.

Virus
Neutralization Results | Tube # |
1 | 2 | 3 | 4 | 5 | 6 | 7 |
8 | 9 | 10 |
| Serum dilution |
1:2 | 1:4 | 1:8 | 1:16 | 1:32 | 1:64 |
1:128 | Ab
Control | RBC
Control | Virus
Control |
Neutralization
Results (Acute)
|
|
|
|
|
|
|
|
|
|
|
Neutralization
Results (Conval.) |
|
|
|
|
|
|
|
|
|
|
VIRUS
NEUTRALIZATION TITER: ACUTE______ CONVAL.______
Was there a fourfold or
greater difference in titers? Virus Neutralization
Results
IMMUNOLOGICAL TERMS - ANTIBODY:
- A globular
protein that is produced by lymphocytes and plasma cells in response to an
antigenic stimulus and that reacts specifically with the eliciting antigen.
- IMMUNOGLOBULIN - A general term for all globulin fractions that contain
antibody.
- ANTIGEN BINDING SITE - That portion of the antibody molecule that
binds to the antigenic determinant on the antigen. It is composed of the
variable amino acid sequences of both light and heavy chains. One light chain
and one heavy chainare required to form a single antigen-combining site.
- HETEROGENEITY OF ANTIBODY - The concept that many different kinds of antibody
can be produced in response to a single antigen. Antibodies to a single antigen
can differ from one another with respect to their antigen binding site, as well
as physical, chemical and biological properties.
- ANTIGEN:
- Any substance (usually foreign) that can initiate an immune
response and on subsequent exposure can react specifically with the product(s)
of that immune response.
- IMMUNOGEN -A more precise term to denote an
antigen that in one physical or chemical form may produce a more pronounced
immune response than inanother form.
- TOLEROGEN - An antigen that invokes a
specific immunological unresponsiveness due to its physical or molecular form. A
tolerogen can become an immunogen ifits molecular form is changed.
- HAPTEN - A
simple chemical group of low molecular weight, that by itself cannot elicit an
immune response, but can do so if combined with a protein carrier.However, the
hapten alone can react with the antibody elicited by the hapten- carrier complex
(in e.g., hapten inhibition).
- ANTIGENIC DETERMINANT -That portion of the
whole antigen molecule that actually combines with antibody or sensitized "
T" lymphocytes.
- ANTIGEN-ANTIBODY REACTIONS:
- Antibody
in solution binds to its specific antigen. This can result in various reactions,
depending on the physical state and natureof the antigen.
- PRECIPITATION -
Soluble antigen and antibody in optimum proportion to one another can form
insoluble complexes that precipitate out of solution.
- AGGLUTINATION - Antigen
bound to (or an integral part of) the surface of an insoluble particle (e.g.,
mammalian or bacterial cell) can bind with its specific antibody to cause
aggregation of the particles.
- NEUTRALIZATION - Antigens that have enzymatic
or toxic properties can react with antibody and as a result be neutralized or
rendered inactive. For example,the interaction of viruses and antibody will
prevent the virus from attaching and entering the host' s cells, thus
neutralizing viral infectivity.
- CELL MEDIATED IMMUNITY (CMI):
- A
part of the immune response to an antigen that involves the production of
specifically primed " T" lymphocytes. These cellsare
involved in the rejection of tumors and transplanted tissue as well as in defense
against certain bacterial, mycotic, parasitic, and viral infections.
- COMPLEMENT:
- A complex set of serum proteins that are activated when
antibody binds to antigen, by soluble antigen-antibody complexes, or by a "
by-pass" system (alternate pathway) in the absence of specific
antibody/antigen interactions.
- CYTOLYSIS:
- The lysis of bacteria or tumor
or red blood cells by insertion of the membrane attack complex derived from
complement activation.
- HISTOCOMPATIBLE:
- Sharing transplantation
antigens.
- HYPERSENSITIVITY REACTIONS:
- These occur in 4 types:
- ANTIBODY-MEDIATED HYPERSENSITIVITY
- Type I - Anaphylactic ("
Immediate" ): IgE antibody is induced by allergen and binds to mast
cells and basophils. After encountering the antigen again, the fixed IgE becomes
cross-linked, inducing degranulation and release of mediators, especially
histamine.
- Type II - Cytotoxic: Antigens on a cell surface combine with
antibody, that leads to complement-mediated lysis or other cytotoxic membrane
damage.
- Type III - Immune complex: Antigen-antibody immune complexes are
deposited in tissues, complement is activated, and PMNs are attracted to the
side, causing tissue damage.
- CELL-MEDIATED HYPERSENSITIVITY
- Type IV - Delayed: T lymphocytes, sensitized by an antigen, release
lymphokines upon second contact with the same antigen. The lymphokines induce
inflammation and activate macrophages.
- IMMUNE RESPONSE:
- The immunological response of a host exposed to a
foreign substance (antigen); it can be humoral (antibody production) and/or cell-
mediated (sensitized " T" lymphocytes).
- "
T" LYMPHOCYTE - A " thymic dependent" small
lymphocyte that is responsiblefor the cell-mediated immune response and that
recognizes antigen and influences production of antibody by interaction with
" B" lymphocytes.
- " B" LYMPHOCYTE -
A " thymic independent" (bone marrow?) small lymphocyte
characterized by surface immunoglobulin antigen-receptors. It is thought to
differentiate into plasma cells that produce antibody.
- AUXILIARY CELL -
Macrophage or blood monocyte that " presents" antigen to
" T" and " B" lymphocytes in a
nonspecific manner for the initiation of an immune response.
- PLASMA CELL - A
highly specialized " end" cell that synthesizes large
amounts of antibody.
- IMMUNITY:
- A classical state of increased protection against or reaction
to the effects of a foreign substance or microorganism involving all parameters
of the immune response.
: - NATURAL IMMUNITY - Nonspecific resistance not
acquired through contact with an antigen. It includes skin and mucous membrane
barriers and a variety of nonspecific immunologic factors.
- ACQUIRED IMMUNITY
- Protection acquired by natural or deliberateintroduction of an antigen into a
responsive host. It is specific and is mediated by eitherantibody or lymphoid
cells (or both).
- MEMBRANE ATTACK COMPLEX:
- The end product of the
activation of the complement cascade. MAC makes holes in the membranes of
gram-negative bacteria, killing them, and in red blood or other cells, causing
them to lyse.
- OPSONIZATION:
- The coating of an antigen or particle by
substances (called opsonins) such as antibodies, complement components, etc.,
that facilitate uptake of the foreign particle into a phagocytic cell.
- PHAGOCYTOSIS:
- The process of internalization of particulate matter by
cells such as macrophages and polymorphonuclear leukocytes (PMNs).
While every effort is made to ensure that this information is up-to-date and accurate, the statements found on this page are for informational purposes only.LSUMC WWW Publication Policy, 9/23/96





